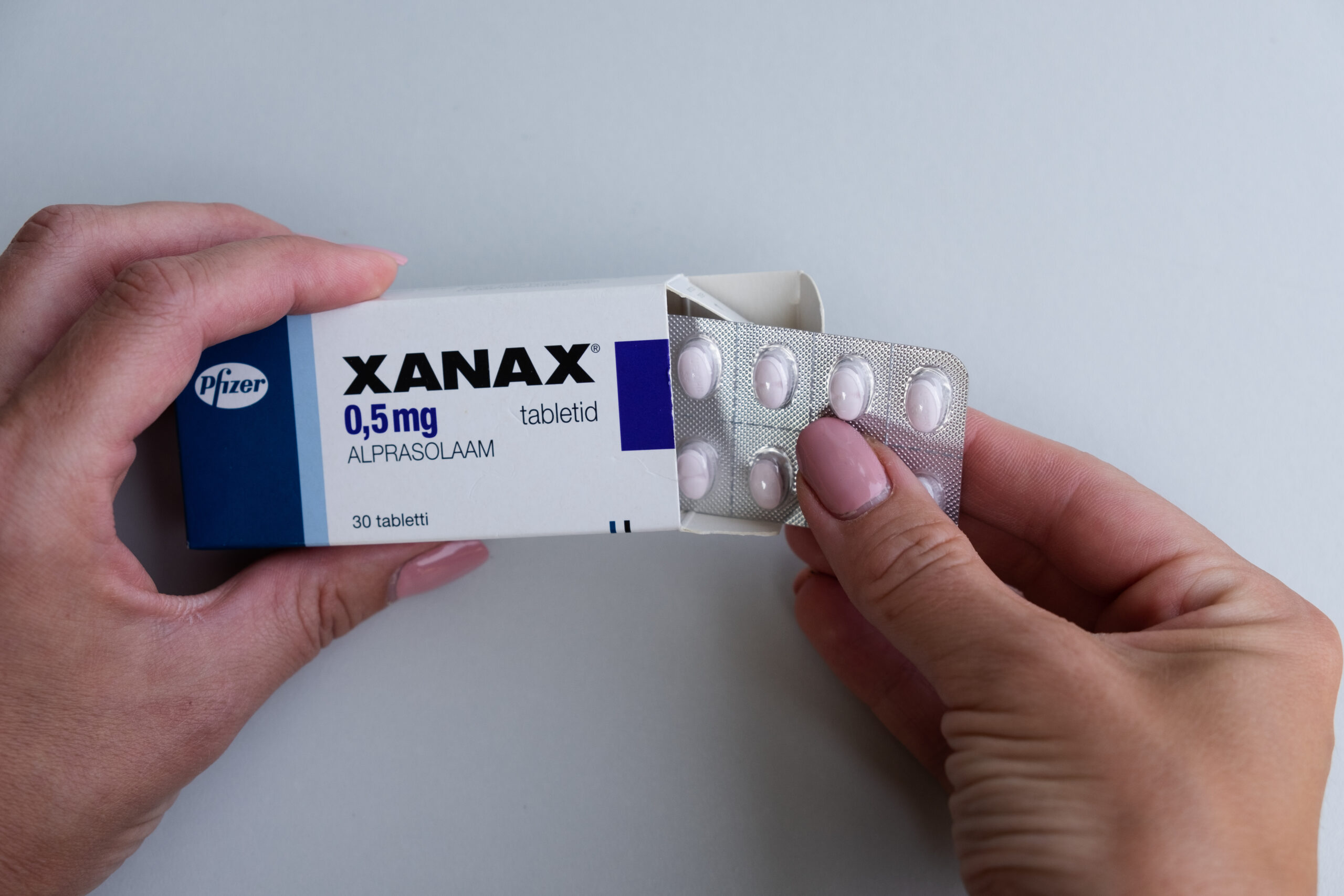
A nationwide recall of Xanax XR has been issued, and if you or someone you know takes this medication for anxiety or panic disorder, there is one specific thing to check right now: the lot number on your bottle.
The recall is real, it covers the entire United States, and it was escalated to a formal FDA classification on April 8, 2026.
It is also a recall with meaningful limits that most headlines have not made fully clear, and understanding those limits is as important as knowing the recall exists.
Here is everything confirmed so far.
What Is Being Recalled?
The product under recall is Xanax XR, the extended-release formulation of alprazolam, the anxiety and panic disorder medication.
The recall was initiated by Viatris Specialty LLC, the pharmaceutical company that manufactures and distributes Xanax XR.
Viatris first initiated the recall on March 17, 2026. The FDA classified it as a Class II recall on April 8.
The recall covers one specific lot. Only one. The lot number is 8177156. The bottles affected are 3 mg extended-release tablets in 60-tablet bottles, available by prescription only.
The expiration date printed on the affected bottles is February 28, 2027.
That lot was distributed throughout the United States between August 27, 2024 and May 29, 2025.
If your bottle of Xanax XR does not have lot number 8177156 on it, it is not part of this recall.
The recall does not affect immediate-release Xanax or alprazolam tablets. It does not affect other dosage strengths. It does not affect every bottle of Xanax XR on shelves or in medicine cabinets, only this specific lot.
The number of bottles included in the recalled lot has not been disclosed by the FDA or Viatris.
Why Was Xanax Recalled?
The reason for the recall is described as “failed dissolution specifications.” This is a quality control failure, not a contamination issue. Understanding what dissolution means in pharmaceutical terms matters here.
Dissolution testing is the process pharmaceutical manufacturers use to verify that a drug dissolves and releases properly when it enters the body.
The test simulates what happens in the stomach and bloodstream, it checks whether the medication breaks down at the right rate and releases its active ingredient as intended.
For extended-release medications specifically, this is especially critical. An extended-release tablet is engineered to dissolve slowly over time, delivering medication at a controlled rate rather than all at once.
If the dissolution profile is off, if the tablet dissolves too quickly or too slowly, or releases an inconsistent amount, the patient may receive too much medication in a short window, too little over time, or an unpredictable amount that does not match what was prescribed.
The California State Board of Pharmacy described the recall as being done “out of an abundance of caution” after testing produced dissolution results outside the required specifications.
Viatris did not issue a formal press release about the recall. The company distributed notification letters to pharmacists and healthcare providers. There have been no reports of adverse reactions related to the recalled lot as of the recall notice.
What Does A Class II Recall Mean?
The FDA classifies drug recalls into three categories based on the potential for harm.
A Class I recall is the most serious, it covers situations where continued use of the product could cause serious adverse health consequences or death. A Class III recall is the least serious, it covers situations where the product is unlikely to cause any adverse health consequences at all.
A Class II recall, the classification given to this Xanax XR lot, is the middle category.
It applies to situations where “use of or exposure to a violative product may cause temporary or medically reversible adverse health consequences, or where the probability of serious adverse health consequences is remote.”
A Class II classification means the FDA has determined there is a possibility of harm, but that any harm is expected to be temporary and reversible rather than permanent or life-threatening.
It is not a dismissal of the issue, but it is also not the most urgent category of recall.
What To Do If You’re Affected
The first step is to check your bottle. Look for the lot number, which is typically printed on the bottom or side of the prescription bottle.
If it reads 8177156 and your expiration date is February 28, 2027, your bottle is part of the recall.
If you have the recalled lot, the FDA and the recall notice advise you to contact your pharmacist or your prescribing physician.
You can also contact Viatris directly at (800) 796-9526 or via email at customer.service@viatris.com for guidance on the recall.
What you should not do, and this is important, is stop taking the medication abruptly without first speaking to your doctor or pharmacist.
Alprazolam is a benzodiazepine, and benzodiazepines cause physical dependence with regular use. Stopping a benzodiazepine suddenly without medical supervision can cause serious withdrawal symptoms including anxiety rebound, insomnia, tremors, and in severe cases, seizures.
The recall notice does not suggest discontinuing the medication without guidance, and neither do any of the medical organizations commenting on this recall.
If you have the affected lot, call your pharmacist or doctor and ask what they recommend for your specific situation.
They can advise whether to continue the current supply, switch to a different lot, or adjust your prescription.
Is Xanax Being Pulled From The Market?
Xanax XR as a drug is not being pulled from the market. Alprazolam is not being recalled broadly.
This recall covers a single lot of a single dosage strength in a single bottle size, 3 mg extended-release tablets in 60-count bottles, lot number 8177156, distributed between mid-2024 and mid-2025.
It does not mean that every Xanax XR prescription currently being filled is affected.
It does not mean that immediate-release alprazolam or generic alprazolam tablets are under recall. It is a targeted, lot-specific quality control action for a medication that failed a dissolution test.
What Is Xanax XR?
Xanax XR, alprazolam extended-release, is a benzodiazepine medication prescribed primarily to treat panic disorder and generalized anxiety disorder in adults.
It is also used for depression-related anxiety.
The “XR” formulation is designed to release the medication slowly over an extended period, maintaining more consistent blood levels than the immediate-release version, which acts more quickly and wears off sooner.
Alprazolam is one of the most widely prescribed benzodiazepines in the United States.
Research published in the journal Nature in 2024 found that approximately 56% of alprazolam users are female, with the highest concentration of use among adults between ages 18 and 45.
Use has also risen among older Americans over portions of the past decade.
Viatris, the company issuing this recall, is a major global pharmaceutical company formed in 2020 from the merger of Mylan N.V. and Pfizer’s Upjohn division.
Alprazolam was originally developed by Pfizer and sold under the Xanax brand name, which Viatris now holds.
This is not the first time this specific product has been recalled for the same reason.
In 2022, Viatris recalled a different lot of Xanax XR 3 mg 60-count bottles, lot number EH8348, also for failed dissolution specifications. That recall affected approximately 6,789 bottles and was terminated by the FDA in June 2023.
The Bottom Line
Check the lot number on your Xanax XR bottle. If it says 8177156, contact your pharmacist or doctor before making any changes to how you take it.
Do not stop the medication suddenly. If you have questions, call Viatris at (800) 796-9526.
If your lot number is different, your medication is not affected by this recall.
If you are experiencing any symptoms that you believe may be related to your medication, anxiety that feels less controlled than usual, changes in how the medication seems to be working, that is always worth a conversation with your prescribing physician regardless of whether your lot is affected.