A clinical trial published in the New England Journal of Medicine in May 2026, and supplemented by Phase 3 data announced in April that nearly doubled survival in patients with advanced pancreatic cancer, has made daraxonrasib the most consequential development in pancreatic cancer treatment in a generation.
Scientists once believed the genetic mutation that drives more than 90 percent of pancreatic cancers could never be targeted with a drug. That belief has now been overturned.
“This drug is potentially going to be a landmark shift in how we treat pancreatic cancer,” said Dr. Eileen O’Reilly of Memorial Sloan Kettering Cancer Center, who led the Phase 1/2 clinical trial of daraxonrasib. “The ability to target RAS mutations that drive so much of pancreatic cancer is huge and has been missing from our treatments.”
The Phase 3 trial result, announced by Revolution Medicines on April 13, 2026, reported that patients with previously treated metastatic pancreatic cancer who received daraxonrasib survived a median of 13.2 months, compared to 6.7 months for patients receiving standard chemotherapy.
The full data will be presented in a plenary session at the American Society of Clinical Oncology Annual Meeting on May 31, 2026.
Why Pancreatic Cancer Is So Difficult To Treat
Pancreatic cancer sits at the brutal end of the spectrum in oncology. The disease is projected to become the second most deadly cancer in the United States by 2030, and the statistics that describe its current trajectory explain why that projection is not a surprise.
Most patients are diagnosed at an advanced stage, when the cancer has already spread.
The five-year survival rate for all pancreatic cancer patients combined is approximately 12 to 13 percent, significantly lower than breast cancer, prostate cancer, colon cancer and most other cancers that receive comparable research attention.
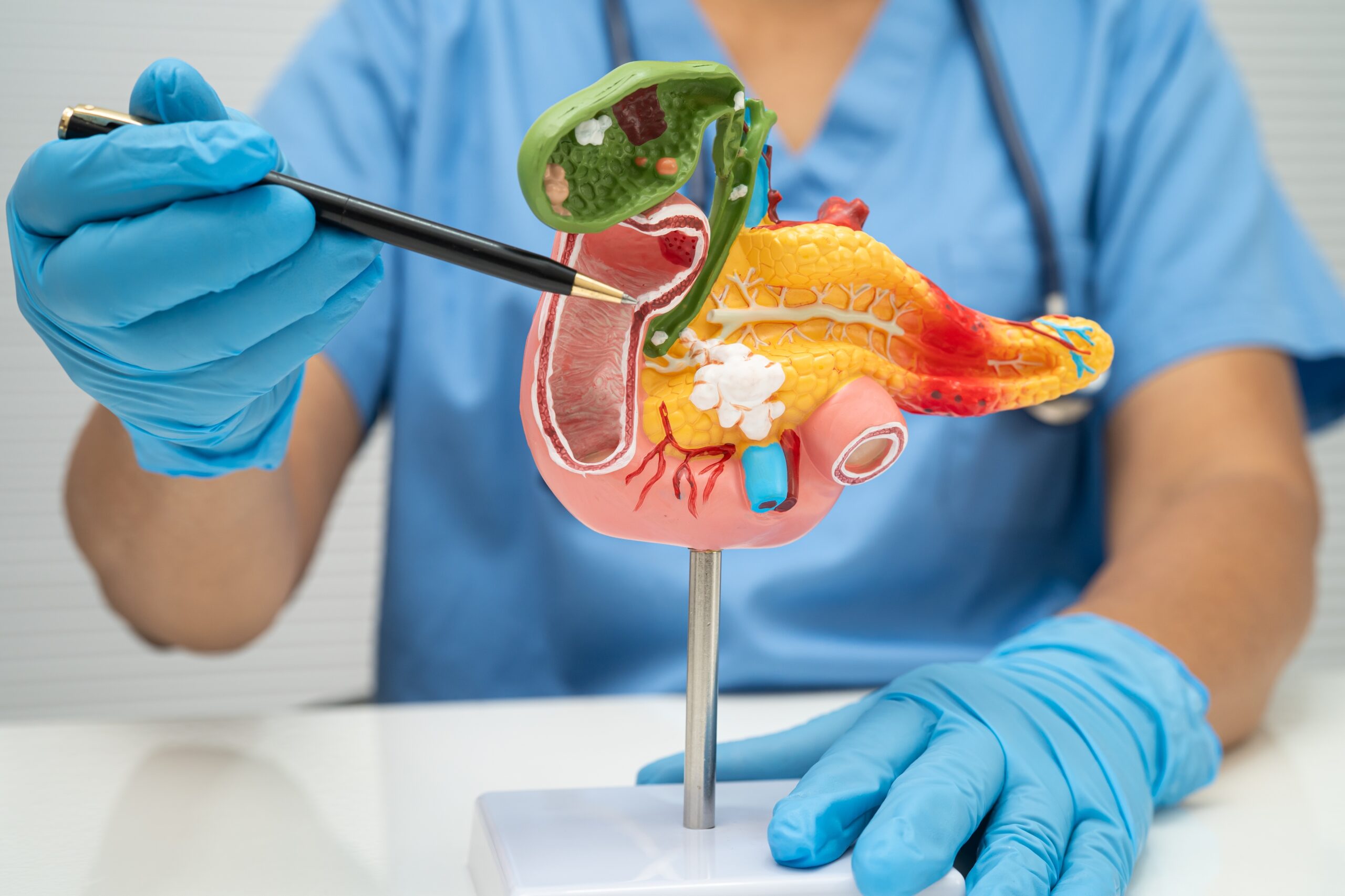
The primary biological driver of pancreatic cancer has been known for decades.
More than 90 percent of pancreatic tumors carry a mutation in the KRAS gene, a gene that normally helps regulate when cells grow and divide. When KRAS mutates, it becomes stuck in an “on” position, sending constant signals to cells telling them to keep dividing.
Those cells “just keep dividing and growing and dividing until it practically snuffs out the life of the patient,” said Dr. Olatunji Alese, an oncologist at Emory University’s Winship Cancer Institute who specializes in gastrointestinal cancers.
For decades, oncologists knew that KRAS was the driver. They could not do anything about it.
The KRAS protein has a smooth surface with few obvious places where a drug molecule can bind, it was described in the research literature as “undruggable,” a cancer biology term meaning the structural characteristics of the protein make it essentially inaccessible to conventional drug design.
Researchers kept trying. The investments were enormous. The failures were consistent. The mutation that drives nine out of ten pancreatic tumors could not be touched.
Daraxonrasib is the drug that changes that.
What Is Daraxonrasib?
Daraxonrasib is a RAS(ON) inhibitor, specifically a pan-RAS inhibitor, meaning it can target multiple forms of the RAS family of proteins rather than just one specific mutation variant.
It is manufactured as an oral pill taken once daily rather than an intravenous chemotherapy that requires a hospital or clinic visit for administration.
For patients managing a cancer diagnosis while trying to maintain quality of life, the difference between a daily pill and a scheduled IV infusion is not a minor detail.
The drug works by binding directly to mutant RAS proteins, the altered versions of KRAS and related proteins that drive uncontrolled cancer cell growth.
By binding to those proteins, daraxonrasib blocks the signals they send. The cells that were receiving constant “keep dividing” instructions from the mutant KRAS no longer receive them.
For many patients in the clinical trials, the effect has been rapid and visible.
“Some patients reported that their symptoms improved just days after taking the drug,” said Dr. Whijae Park of Memorial Sloan Kettering, who served as the pancreatic cancer lead for the Phase 1/2 study. “We do not see that quick response from most chemotherapy regimens.”
Dr. Park’s observation is consistent with what targeted therapies have shown in other cancers where a driver mutation can be blocked directly. When you remove the signal that was powering the tumor’s growth, the tumor’s response can be faster and more dramatic than what chemotherapy, which works by killing rapidly dividing cells broadly rather than targeting a specific molecular driver, typically produces.
The Clinical Trial Results That Changed The Conversation
The phase 1/2 study of daraxonrasib in patients with previously treated advanced pancreatic cancer was published in the New England Journal of Medicine in May 2026, the most prestigious medical journal in the world, and the publication venue that signals a finding has been vetted and validated to the standard that determines whether the broader medical community accepts a result as credible.
The trial involved 16 cancer centers across the United States. At the 300 milligram daily dose, the dose that was subsequently selected for Phase 3 testing, approximately 30 percent of patients with one prior line of treatment experienced an objective response, meaning their cancer measurably shrank.
Approximately 90 percent of all patients in the trial experienced disease control, meaning their cancer was either reduced or stabilized rather than continuing to progress.
The median duration of response for patients with one prior therapy was more than 8 months.
For context: the standard of care for previously treated metastatic pancreatic cancer involves chemotherapy regimens that produce objective response rates significantly below what the daraxonrasib trial achieved.
The drug was also generally better tolerated than chemotherapy, patients described fewer and more manageable side effects, which is consistent with the biology of targeted therapy versus broad-spectrum cytotoxic treatment.
The Phase 3 RASolute 302 trial, which enrolled patients across a global study design, produced the numbers that sent the oncology community’s pulse rate upward when Revolution Medicines announced them on April 13, 2026.
In the full patient population, all enrolled patients with previously treated metastatic pancreatic ductal adenocarcinoma, daraxonrasib produced a median overall survival of 13.2 months against 6.7 months for investigator’s choice chemotherapy.
The hazard ratio was 0.40, with a p value less than 0.0001, statistical language meaning the result was so unlikely to occur by chance that the probability of it being random is effectively zero.
A hazard ratio of 0.40 means patients on daraxonrasib had 40 percent of the risk of death at any given time compared to patients receiving chemotherapy.
The median survival gain, 13.2 months versus 6.7 months, nearly doubled the time patients lived.
For a disease where the historically expected survival in this setting was measured in months rather than years, doubling it is the kind of result that oncologists describe as transformative.
“The widely anticipated results of this study indicate that daraxonrasib provides a clear and highly meaningful benefit for patients with metastatic pancreatic cancer,” said Dr. Brian Wolpin, professor of medicine at Harvard Medical School, director of the Hale Family Center for Pancreatic Cancer Research at Dana-Farber Cancer Institute and the principal investigator for the RASolute 302 trial.
The FDA’s Role And What Comes Next
Revolution Medicines has not yet applied to the FDA for approval of daraxonrasib.
That application, called a New Drug Application, is the step between completed clinical trials and an approved drug that doctors can prescribe.
The company has stated its intention to submit the Phase 3 data to global regulatory authorities including the FDA as part of a future New Drug Application.
The regulatory path for daraxonrasib has several features that suggest the FDA will move with unusual speed.
The agency awarded the drug “Breakthrough Therapy” designation in June 2025, based on encouraging early results from the Phase 1 trial. Breakthrough Therapy designation is a specific FDA program that accelerates the development and review process for drugs that show substantial improvement over existing treatments for serious conditions.
In October 2025, the FDA selected daraxonrasib for its “Commissioner’s National Priority Voucher” pilot program, an initiative to accelerate review of therapies aligned with national health priorities, a category pancreatic cancer clearly fits given its mortality burden and the absence of effective targeted treatments.
In May 2026, the FDA issued a “safe to proceed” letter allowing the drug to be administered to additional patients in ongoing testing.
The full Phase 3 results will be presented at the plenary session of the ASCO Annual Meeting on May 31, 2026.
ASCO plenary presentations are the highest-visibility slot at the most important oncology conference in the world, reserved for trial results that the conference’s scientific committee judges to be practice-changing findings in cancer medicine.
The selection of the RASolute 302 results for plenary presentation at ASCO signals that the oncology community’s leadership views this as exactly that.
What The Researchers Say About Its Significance
The language that researchers and oncologists have used to describe daraxonrasib is not the cautious hedge language that normally accompanies early drug results.
Normally, cancer researchers discuss promising findings with significant qualifiers about the need for more data, larger populations and longer follow-up.
Those qualifiers exist here too, Phase 3 is not FDA approval, and FDA approval is not full clinical adoption.
But the characterizations being offered by respected researchers at MSK, Dana-Farber and other leading institutions point to a consensus that this result is genuine and significant.
“I think this is truly an earth-shattering moment in pancreatic cancer,” one researcher told National Geographic. Dr. O’Reilly called it “potentially a landmark shift.” Dr. Wolpin’s statement described the benefit as “clear and highly meaningful.”
The Pancreatic Cancer Action Network described it as extending survival “for the first time” with a RAS inhibitor.
These are not the descriptions of researchers tempering expectations ahead of uncertain results. They are descriptions of researchers who know what they are looking at and what it means.
The mutation that drives pancreatic cancer was called undruggable. The drug that targets it is now going through FDA review with the full Phase 3 data in hand.
Patients who receive a metastatic pancreatic cancer diagnosis today are still facing one of the most difficult prognoses in oncology. Patients who receive that diagnosis after daraxonrasib is approved may be facing something different.